(volume in liter * pressure in BAR) = content in liter
Unfortunately air does not quite follow this simple equation. Especially when dealing with pressure as high as 200 BAR (3000 PSI) or above. In general, when using this equation, the higher the pressure gets, the more inaccurate the result gets. That's why we need to add the compressibility factor of air, to the equation, which will give us a much more realistic result. The equation then look like this:
(volume in liter * pressure in BAR) / compressibility factor = content in liter
The compressibility factor of air is a way of describing how much air "resists" being compressed at a given pressure and temperature. So in order to find the compressibility factor of air we only need to know one more thing: the temperature.
The green line in the graph below, shows the compressibility factor of dry atmospheric air at 25°C from 0 to 500 BAR.
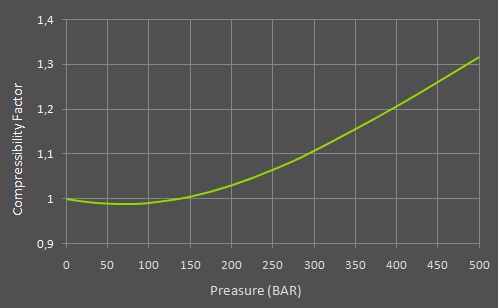
The need for compressibility factor is probably best visualized using a small example:
Let's say we have a tank with an internal volume of 1 liter (1000cc). We fill this tank to 1 BAR, and use the equation without the compressibility factor, to calculate the content. It will look like this:
(1 liter * 1 BAR) = 1 liter
Very simple, 1 liter of air in the tank. We fill the tank to 2 BAR and we will have: (1 liter * 2 BAR) = 2 liters of air in the tank. 3 BAR = 3 liters, 100 BAR = 100 liters, 300 BAR = 300 liters and so on.
Now let's try and calculate the content of air, in the same small tank, using the equation with the added compressibility factor of air. We'll fill it to 300 BAR again, on a nice warm day with a temperature of 25°C (77°F). First we need to find the specific compressibility factor of air for 300 BAR in the graph above, which is 1.1074. We then add this factor to the equation, and our calculation will look like this:
(1 liter * 300 BAR) / 1.1074 = 270.9 liters
Calculating the content without the compressibility factor, we would have gotten 300 liters. So what happened to the remaining 29.1 liters of air? Well, the 29.1 liters just never got into the tank before the pressure reached 300 BAR. This is because air with a temperature on 25°C at 300 BAR "resists" being compressed that much more. Making us reach 300 BAR of pressure, 29.1 liters of air short, so to speak.
29.1 liters might not sound of much, but remember that this is a tank with a volume on only 1 liter.
If we do the same calculation with a quite common scuba tank, with a 12 liter volume filled to 300 BAR (4351 PSI), we would see that almost 350 liters would be "missing", compared to not using the compressibility factor when calculating the content.
350 liters of air could easily be worth as much as 30 refills of an air gun, depending of the needed pressure and tank size. So do yourself a favor, and make a few calculations before buying a scuba tank for your air gun - So you know what you can expect.